|
The symbol K originates from the Latin word kalium. The origin of the element's name comes from the English word 'potash,' meaning pot ashes, and the Arabic word qali, which means alkali. In its elemental form, potassium has a silvery gray metallic appearance, but its compounds (such as potassium hydroxide) are more frequently used in industrial and chemical applications. As with other alkali metals, potassium decomposes in water with the evolution of hydrogen because of its reacts violently with water, it only occurs in nature in ionic salts. It is one of the most reactive and electropositive of all metals and rapidly oxidizes. Potassium is the seventh most abundant element on earth. Potassium was discovered and first isolated by Sir Humphrey Davy in 1807. The potassium atom has a radius of 227.2 pm and a Van der Waals radius of 275 pm. The number of electrons in each of Potassium's shells is and its electron configuration is 4s 1. Potassium (atomic symbol: K, atomic number: 19) is a Block S, Group 1, Period 4 element with an atomic weight of 39.0983. Additional technical, research and safety (MSDS) information is available as is a Reference Calculator for converting relevant units of measurement. Typical and custom packaging is available. American Elements produces to many standard grades when applicable, including Mil Spec (military grade) ACS, Reagent and Technical Grade Food, Agricultural and Pharmaceutical Grade Optical Grade, USP and EP/BP (European Pharmacopoeia/British Pharmacopoeia) and follows applicable ASTM testing standards. Metallic ions can also be dispersed utilizing suspended or coated nanoparticles and deposited utilizing sputtering targets and evaporation materials for uses such as solar cells and fuel cells. Organometallic forms are soluble in organic solutions and sometimes in both aqueous and organic solutions. Most metal sulfate compounds are readily soluble in water for uses such as water treatment, unlike fluorides and oxides which tend to be insoluble. Sulfate compounds are salts or esters of sulfuric acid formed by replacing one or both of the hydrogens with a metal. High purity, submicron and nanopowder forms may be considered. Thin Film Deposition & Evaporation MaterialsĬhromium Potassium Sulfate is generally immediately available in most volumes.Additive Manufacturing & 3D Printing Materials.Neither ILO nor WHO nor the European Commission shall be responsible for the interpretation and use of the information contained in this material. The published material is being distributed without warranty of any kind, either expressed or implied. The substance is toxic to aquatic organisms.Īll rights reserved. TLV: (as Cr(III), inhalable fraction): 0.003 mg/m 3, as TWA (DSEN) (RSEN) A4 (not classifiable as a human carcinogen). The chemical analysis data show that the chromium and chlorine incorporated in the glass have strong negative effects on sulfur solubility, which can be qualitatively explained by the sulfur, chlorine, and chromium ions competing for voids in the glass matrix. Repeated or prolonged contact may cause skin sensitization. The substance is irritating to the respiratory tract.Ī harmful concentration of airborne particles can be reached quickly when dispersed.Įffects of long-term or repeated exposure 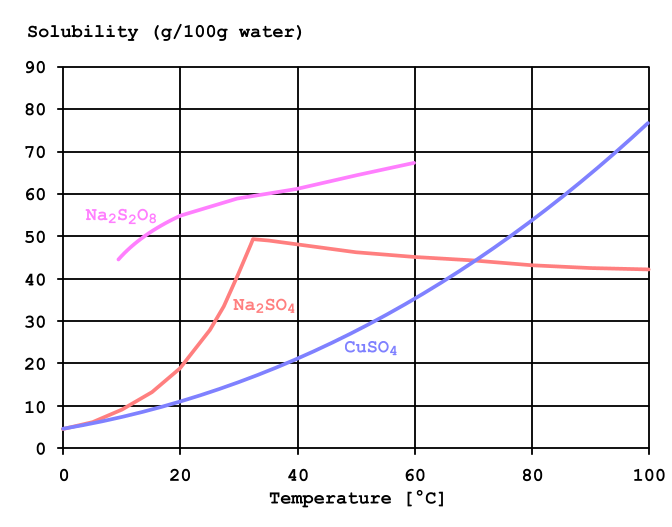
With the financial assistance of the European Commission.ĭecomposes on heating. Prepared by an international group of experts on behalf of ILO and WHO, 
If appropriate, moisten first to prevent dusting. Sweep spilled substance into covered containers. Vacuum spilled material with specialist equipment. Do NOT let this chemical enter the environment. The remaining solution contains chromium sulfate. Specifications Solubility Information, Soluble in water. Personal protection: particulate filter respirator adapted to the airborne concentration of the substance. Soluble fats were removed by solvent extraction using 1:1 mixture of petroleum ether and isopropanol at 60C. The pathway of the sparingly soluble salt can be easily monitored by x-rays. Its solubility product is 1.08 × 10 10 at 25C, so it is ideally suited for this purpose because of its low solubility when a barium milkshake is consumed by a patient. 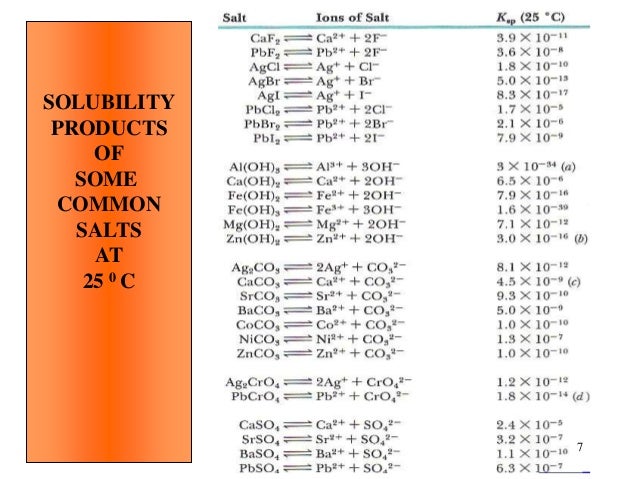 
Give one or two glasses of water to drink. We mentioned that barium sulfate is used in medical imaging of the gastrointestinal tract. Rinse skin with plenty of water or shower.įirst rinse with plenty of water for several minutes (remove contact lenses if easily possible), then refer for medical attention. Use local exhaust or breathing protection. In case of fire in the surroundings, use appropriate extinguishing media.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
